Monday, February 27, 2012
how to used pressed pellet
Sunday, July 17, 2011
Toxic Heavy Metals
Toxic Heavy Metals
(Source: caobisco)
1. Lead (Pb)
· The main sources of lead pollution in the environment are: Industrial production processes and their emissions, road traffic with leaded petrol, the smoke and dust emissions of coal and gas-fired power stations, the laying of lead sheets by roofers as well as the use of paints and anti-rust agents.
· Lead can trigger both acute and chronic symptoms of poisoning. Acute intoxications only occur through the consumption of relatively large single doses of soluble lead salts.
· In 1993, the joint FAO/WHO Expert Committee for Additives and Contaminants (JECFA) reduced the value it had provisionally specified for adults in 1972, for tolerable lead consumption per week (PTWI - provisional tolerable weekly intake), from 0.05 mg/kg body weight to 0.025 mg (or 25 µg).
· The lead content in drinking water presents a problem in many countries. Thus, the present WHO guideline of 50 µg/l is exceeded in Great Britain, for example, in 34 % of households.
2. Cadmium (Cd)
· Cadmium (world production in 1972, 15 000 t) exists in low concentrations in all soils. It is actively extracted from its ores for commercial purposes and is also emitted in industrial processes such as metal melting and refining, coal and oil-fired power stations, electroplating plants, etc.
· Cadmium is today regarded as the most serious contaminant of the modern age. It is absorbed by many plants and seacreatures and, because of its toxicity, presents a major problem for foodstuffs. Contamination through fertilisers becomes an increasing problem.
· Unlike lead, cadmium contamination cannot be removed from plants by washing them; it is distributed throughout the organism. It is often difficult to be certain of the cause of a cadmium content found in fruit or vegetables, as the substance in its natural form exists everywhere in the soil and is absorbed by the roots.
· Cadmium is concentrated particularly in the kidneys, the liver, the blood forming organs and the lungs. It most frequently results in kidney damage (necrotic protein precipitation) and metabolic anomalies caused by enzyme inhibitions.
· Cadmium, like lead, is a cumulative poison, i.e. the danger lies primarily in the regular consumption of foodstuffs with low contamination.
· In most countries, there are legal regulations regarding permissible cadmium contamination levels. As a rule, these are based on the PTWI value last set by the JECFA of the FAO/WHO in 1989 of about 7 µg/kg body weight, corresponding with a quantity of 0.4-0.5 mg per person (70 kg) per week.
· One problem of a special kind is the smoking of tobacco. 20 cigarettes a day provide a cadmium input of 4 µg.
3. Mercury (Hg)
· Passes into the environment through emissions from chemical plants (paints, paper, chlorine, plant pesticides) and power stations, mostly in effluents and sludge.
· Mercury becomes concentrated in shellfish, crustaceans and fish and thus also passes, in the form of highly toxic mercury methylate, into the human food chain.
· Mercury in the form of its methyl compounds is specifically the most toxic of the heavy metals.
· When consumed orally, it first passes into the liver, the kidneys and the brain.
· In the case of chronic consumption, first cause tiredness, loss of appetite and weight loss. In the end the kidneys fail. Muscular weakness and paralysis are typical.
· The FAO/WHO Expert Committee (JECFA) has set a provisional maximum acceptable value for mercury consumption at 5 µg per kg body weight per week.
4. Arsenic (As) (source)
· Arsenic is the most common cause of acute heavy metal poisoning in adults and is number 1 on the ATSDR's "Top 20 List."
· Arsenic is released into the environment by the smelting process of copper, zinc, and lead, as well as by the manufacturing of chemicals and glasses.
· Arsine gas is a common byproduct produced by the manufacturing of pesticides that contain arsenic.
· Arsenic may be also be found in water supplies worldwide, leading to exposure of shellfish, cod, and haddock. Other sources are paints, rat poisoning, fungicides, and wood preservatives.
· Target organs are the blood, kidneys, and central nervous, digestive, and skin systems (Roberts 1999; ATSDR ToxFAQs for Arsenic).
5. Others Heavy Metals
· Some heavy metals (the so-called trace elements) are essential in very small concentrations for the survival of all life forms, for example, copper, iron, zinc, chromium, molybdenum and others.
· Nickel has often been associated recently with allergies (contact with jewellery and jeans buttons containing nickel). There is no established knowledge of effects of this type when it is absorbed in the gastro-intestinal tract. Cocoa is one of the foodstuffs with higher than average natural nickel contents.
· The copper content of tomato dishes prepared in copper pots and having a copper content of 0.1-0.2 mg/kg body weight has already been found to cause digestive disturbances in sensitive consumers! This is in spite of the specified tolerable quantity for daily consumption of 0.5 mg/kg.
· Chromium, copper and zinc play major roles in modern industry and, in the vicinity of extraction or processing plants, the emissions arising are certainly capable of causing an undesirable contamination of agricultural products. Considerable quantities have been found in fruit and vegetables. However, no adverse effects on health are known. It is nevertheless recommended not to omit these metals a priori from scrutiny.
Monday, June 27, 2011
Ionizing Radiation

Friday, June 10, 2011
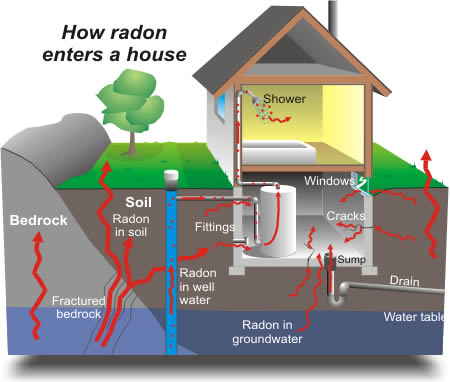
What is Radon
- cracks at concrete floor-wall junctions
- gaps in the floor
- small pores in hollow-block walls
- sumps and drains.
- improving the ventilation of the house
- avoiding the passage of radon from the basement into living rooms
- increasing under-floor ventilation
- installing a radon sump system in the basement
- sealing floors and walls
- installing a positive pressurization or ventilation system.

- Not everyone exposed to even high levels of radon will contract radon induced lung cancer.
- All radon levels can be lowered, which lowers the risk assessment.

